A) 5.3 × 1013
B) 4.9 × 1011
C) 1.4 × 109
D) 7.5 × 107
E) 3.8 × 104
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which electron energy will produce the largest cutoff wavelength for X-ray production from a nickel (Z = 28) surface?
A) 25 keV
B) 30 keV
C) 35 keV
D) 40 keV
E) 45 keV
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the quantum mechanical picture of the atom, which one of the following is a true statement concerning the ground state electron in a hydrogen atom?
A) The ground state electron has zero kinetic energy.
B) The ground state electron has zero binding energy.
C) The ground state electron has zero ionization energy.
D) The ground state electron has zero spin angular momentum.
E) The ground state electron has zero orbital angular momentum.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following list of electron configurations: 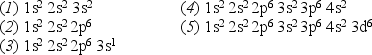 -Which one of the above lists represents the electronic configuration for the ground state of the atom with Z = 11?
-Which one of the above lists represents the electronic configuration for the ground state of the atom with Z = 11?
A) 1
B) 2
C) 3
D) 4
E) 5
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The kinetic energy of the ground state electron in hydrogen is +13.6 eV. What is its potential energy?
A) -13.6 eV
B) +27.2 eV
C) -27.2 eV
D) +56.2 eV
E) zero eV
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A neutral atom has the following electronic configuration: 1s2 2s2 2p6 3s2 3p5 -How many protons are in the atomic nucleus?
A) 4
B) 7
C) 12
D) 17
E) 34
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following statements concerning the cutoff wavelength typically exhibited in X-ray spectra is true?
A) The cutoff wavelength depends on the target material.
B) The cutoff wavelength depends on the potential difference across the X-ray tube.
C) The cutoff wavelength is independent of the energy of the incident electrons.
D) The cutoff wavelength occurs because of the mutual shielding effects of K-shell electrons.
E) The cutoff wavelength occurs because an incident electron cannot give up all of its energy.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Electrons in an X-ray tube are accelerated through a potential difference of 40 kV. The electrons then strike a zirconium (Z = 40) target. Determine the cutoff frequency for X-ray production.
A) 4.7 × 1019 Hz
B) 9.7 × 1018 Hz
C) 3.2 × 1018 Hz
D) 6.7 × 1017 Hz
E) 1.1 × 1016 Hz
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A neutral atom has the following electronic configuration: 1s2 2s2 2p6 3s2 3p5 -How many electrons are in the M shell?
A) 2
B) 5
C) 6
D) 7
E) 8
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following sets of quantum numbers is not possible? 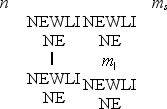
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the energy of the photon emitted when the electron in a hydrogen atom undergoes a transition from the n = 7 level to the n = 4 level.
A) 0.17 eV
B) 0.21 eV
C) 0.36 eV
D) 0.57 eV
E) 1.3 eV
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An electron is in the ground state of a hydrogen atom. A photon is absorbed by the atom and the electron is excited to the n = 2 state. What is the energy in eV of the photon?
A) 13.6 eV
B) 10.2 eV
C) 3.40 eV
D) 1.51 eV
E) 0.54 eV
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete the following statement: For the ground state of the hydrogen atom, the Bohr model correctly predicts
A) only the energy.
B) only the angular momentum.
C) only the angular momentum and the spin.
D) the angular momentum and the energy.
E) the energy, the angular momentum, and the spin.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following statements concerning the plum-pudding model of the atom is false?
A) There is no nucleus at the center of the plum-pudding model atom.
B) The plum-pudding model was proven correct in experiments by Ernest Rutherford.
C) The plum-pudding model was proposed by Joseph J. Thomson.
D) Positive charge is spread uniformly throughout the plum-pudding model atom.
E) Negative electrons are dispersed uniformly within the positively charged "pudding" within the plum-pudding model atom.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following pairs of characteristics of light is best explained by assuming that light can be described in terms of photons?
A) photoelectric effect and the effect observed in Young's experiment
B) diffraction and the formation of atomic spectra
C) polarization and the photoelectric effect
D) existence of line spectra and the photoelectric effect
E) polarization and the formation of line spectra
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
To which model of atomic structure does the Pauli exclusion principle apply?
A) the nuclear atom
B) the quantum mechanical atom
C) the billiard ball atom
D) the plum-pudding model
E) the planetary model
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electrons could be accommodated in an f subshell?
A) 6
B) 8
C) 10
D) 14
E) 18
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete the following statement: In the laser-based medical procedure known as photorefractive keratectomy (PRK) , nearsightedness and farsightedness can be treated using the laser to
A) remove small amounts of tissue from the lens and change its curvature.
B) remove small amounts of tissue from the cornea and change its curvature.
C) change the index of refraction of the aqueous humor.
D) alter the fluid pressure within the eye.
E) stimulate unused rods and cones on the retina.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Two possible states for the hydrogen atom are labeled A and B. The maximum magnetic quantum number for state A is +3. For state B, the maximum value is +1. What is the ratio of the magnitudes of the orbital angular momenta, LA/LB, of an electron in these two states?
A) 1.22
B) 1.73
C) 2.00
D) 2.45
E) 3.46
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following subshells is not compatible with a principle quantum number of n = 4?
A) s
B) p
C) d
D) f
E) g
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 74
Related Exams